The Drug instability can be defined as the ability of the drug or formulation to remain in the fixed specification of identity and purity (i.e. chemical, physical, therapeutical, microbial, toxicological states, etc.) up to some specific period of time into a specific storage condition.
Causes of Instability and their Prevention
Common causes of instability and degradation are:
- Hydrolysis
- Oxidation
- Photolysis
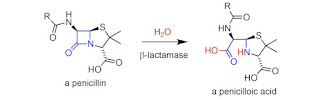
1. Hydrolysis
It is the breaking of molecule due to reaction with water.
Drugs which commonly hydrolysed are: Ester, Ether and Lactams.
A. Hydrolysis of Ester:
B. Hydrolysis of Amide:
Examples: Dibucaine, Ergometrine, Chloramphenicol, Barbiturates, etc.